Varicella Virus Vaccine (live) [Oka/SK]
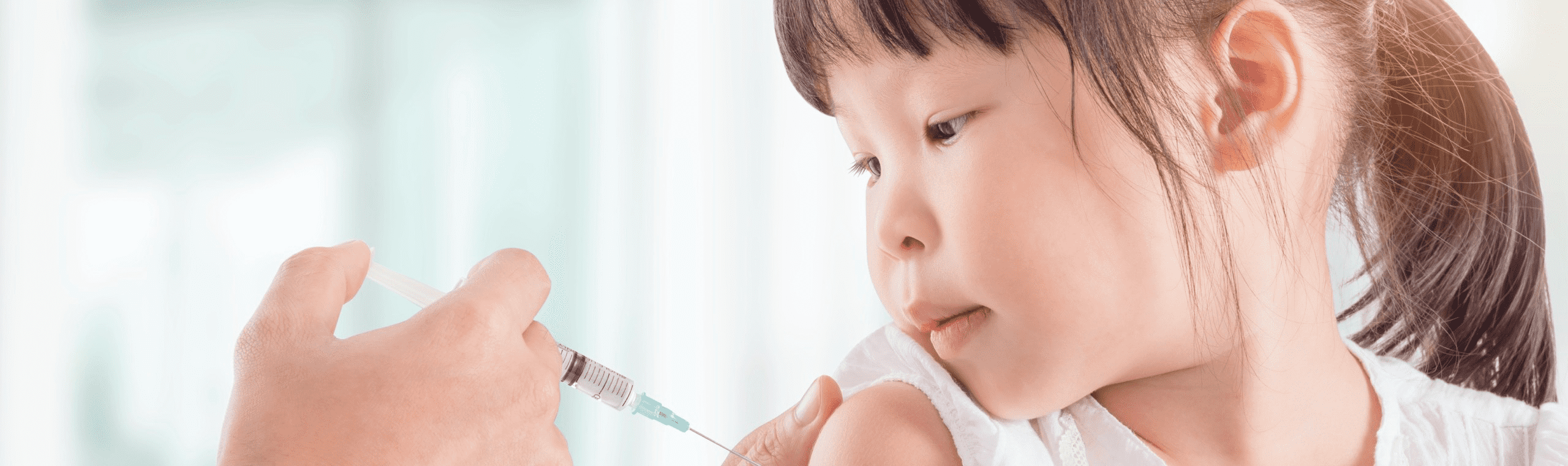
Indication : Prevention of varicella in children 12 months to 12 years of age.
ความถูกต้องของโฆษณานี้เป็นความรับผิดชอบของผู้โฆษณา มิได้ดำเนินการโดยสำนักงานคณะกรรมการอาหารและยา
เลขทะเบียน 1C 23/63 (NBC)
โปรดอ่านรายละเอียดเพิ่มเติมในเอกสารกำกับยา
ฆศ.2-4085/2568
 เป็นยาใหม่ ใช้เฉพาะสถานพยาบาลแพทย์ควรติดตามผลการใช้ยา
เป็นยาใหม่ ใช้เฉพาะสถานพยาบาลแพทย์ควรติดตามผลการใช้ยา
ความถูกต้องของโฆษณานี้เป็นความรับผิดชอบของผู้โฆษณา มิได้ดำเนินการโดยสำนักงานคณะกรรมการอาหารและยา
เลขทะเบียน 1C 23/63 (NBC)
โปรดอ่านรายละเอียดเพิ่มเติมในเอกสารกำกับยา
ฆศ.2-4085/2568
 เป็นยาใหม่ ใช้เฉพาะสถานพยาบาลแพทย์ควรติดตามผลการใช้ยา
เป็นยาใหม่ ใช้เฉพาะสถานพยาบาลแพทย์ควรติดตามผลการใช้ยา
อ่านเพิ่มเติม
(PCEC Rabies Vaccine)
Indication : Active immunization against rabies.
a.Pre-exposure immunisation (preventative, prior to exposure):
Immunisation prior to possible infection with rabies, particularly for vets, veterinary medicine students, animal keepers, hunters, forestry workers, animal handlers, butchers, personnel in rabies research laboratories etc., or prior to visit to areas in which rabies is endemic (rabies infected areas).
b.Post-exposure treatment (after exposure):
Treatment after contact with animals which are rabid or suspected to be rabid, or after contact with an inoculated rabies carcass.
เลขทะเบียน 1C 1/63 (B)
โปรดอ่านรายละเอียดเพิ่มเติมในเอกสารกำกับยา
ฆศ. 2-6784 / 2568
ความถูกต้องของโฆษณานี้เป็นความรับผิดชอบของผู้โฆษณา มิได้ดำเนินการโดยสำนักงานคณะกรรมการอาหารและยา
a.Pre-exposure immunisation (preventative, prior to exposure):
Immunisation prior to possible infection with rabies, particularly for vets, veterinary medicine students, animal keepers, hunters, forestry workers, animal handlers, butchers, personnel in rabies research laboratories etc., or prior to visit to areas in which rabies is endemic (rabies infected areas).
b.Post-exposure treatment (after exposure):
Treatment after contact with animals which are rabid or suspected to be rabid, or after contact with an inoculated rabies carcass.
เลขทะเบียน 1C 1/63 (B)
โปรดอ่านรายละเอียดเพิ่มเติมในเอกสารกำกับยา
ฆศ. 2-6784 / 2568
ความถูกต้องของโฆษณานี้เป็นความรับผิดชอบของผู้โฆษณา มิได้ดำเนินการโดยสำนักงานคณะกรรมการอาหารและยา
อ่านเพิ่มเติม
Influenza vaccine (surface antigen, inactivated, prepared in cell
cultures)
Indication : Active immunization for the prevention of influenza disease caused by influenza virus subtypes A and type B
contained in the vaccine, for adults and children 6 months of age and older.
The use of SKYCellflu Quadrivalent should be based on official recommendations.
เลขทะเบียน 2C 12/66 (NB)
โปรดอ่านรายละเอียดเพิ่มเติมในเอกสารกำกับยา
ฆศ.2-865/2566
ความถูกต้องของโฆษณานี้เป็นความรับผิดชอบของผู้โฆษณา มิได้ดำเนินการโดยสำนักงานคณะกรรมการอาหารและยา
เลขทะเบียน 2C 12/66 (NB)
โปรดอ่านรายละเอียดเพิ่มเติมในเอกสารกำกับยา
ฆศ.2-865/2566
ความถูกต้องของโฆษณานี้เป็นความรับผิดชอบของผู้โฆษณา มิได้ดำเนินการโดยสำนักงานคณะกรรมการอาหารและยา
อ่านเพิ่มเติม
Freeze-dried Live Attenuated Hepatitis A Vaccine
Indication : The vaccine is indicated for active immunization against infection caused by HAV in persons over age of 18 months. It can be used for primary immunization. Hepatitis A vaccine is recommended for pre-exposure prophylaxis of individuals at increased risk of infection and post-exposure prophylaxis. Thus the vaccine is indicated in the following conditions:
1. Residents of communities with high endemic rates or recurrent outbreaks of Hepatitis A;
2. Travellers to countries where Hepatitis A is endemic, especially when the travel involves rural or primitive conditions;
3. Members of the armed forces, emergency relief workers and others likely to be posted abroad at short notice to areas with high rates of HAV infection;
4. Residents and staff of institutions where there is an ongoing problem with HAV transmission;
5. Inmates of correctional facilities in which there is an ongoing problem with HAV infection;
6. People with life style determined risks of infection, including those engaging in oral or intravenous illicit drug use in unsanitary conditions;
7. People with chronic liver disease who may be at increased risk of fulminant hepatits A;
8. Zoo-keepers, veterinarians and researchers who handle non-human primates.
เลขทะเบียน 1C 8/53 (NB)
โปรดอ่านรายละเอียดเพิ่มเติมในเอกสารกำกับยา
ฆศ.2-17/2566
1. Residents of communities with high endemic rates or recurrent outbreaks of Hepatitis A;
2. Travellers to countries where Hepatitis A is endemic, especially when the travel involves rural or primitive conditions;
3. Members of the armed forces, emergency relief workers and others likely to be posted abroad at short notice to areas with high rates of HAV infection;
4. Residents and staff of institutions where there is an ongoing problem with HAV transmission;
5. Inmates of correctional facilities in which there is an ongoing problem with HAV infection;
6. People with life style determined risks of infection, including those engaging in oral or intravenous illicit drug use in unsanitary conditions;
7. People with chronic liver disease who may be at increased risk of fulminant hepatits A;
8. Zoo-keepers, veterinarians and researchers who handle non-human primates.
เลขทะเบียน 1C 8/53 (NB)
โปรดอ่านรายละเอียดเพิ่มเติมในเอกสารกำกับยา
ฆศ.2-17/2566
อ่านเพิ่มเติม
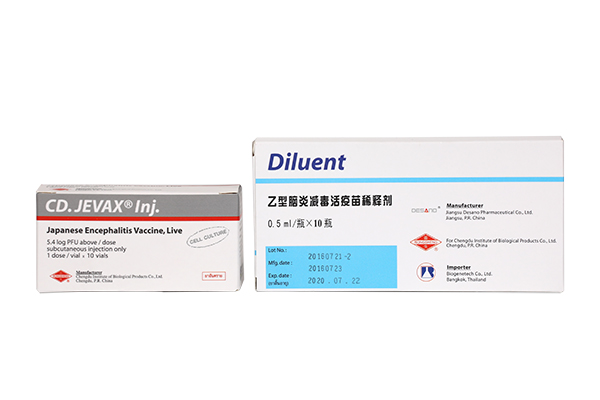
(Japanese Encephalitis Vaccine, Live)
Indication : Prophylactic immunization against Japanese Encephalitis.
เลขทะเบียน 1C 108/50 (NB)
โปรดอ่านรายละเอียดเพิ่มเติมในเอกสารกำกับยา
ฆศ. 2-6785 / 2568
ความถูกต้องของโฆษณานี้เป็นความรับผิดชอบของผู้โฆษณา มิได้ดำเนินการโดยสำนักงานคณะกรรมการอาหารและยา
เลขทะเบียน 1C 108/50 (NB)
โปรดอ่านรายละเอียดเพิ่มเติมในเอกสารกำกับยา
ฆศ. 2-6785 / 2568
ความถูกต้องของโฆษณานี้เป็นความรับผิดชอบของผู้โฆษณา มิได้ดำเนินการโดยสำนักงานคณะกรรมการอาหารและยา
อ่านเพิ่มเติม
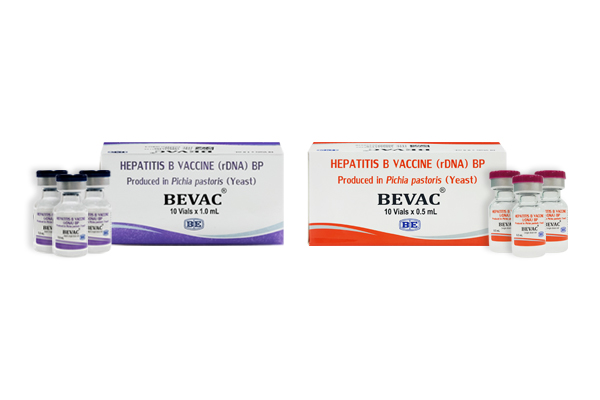
(Hepatitis B Vaccine (rDNA))
Indication : BEVAC is indicated for immunization of persons exposed to infection by hepatitis B virus and its common sub-types. It can also be given to hepatitis C and D virus infected patients to protect them against co-infection with hepatitis B virus. BEVAC is recommended primarily for neonates, infants, children and adults not only for the prevention of the disease but also to protect them from probable hepatitis B virus – induced carrier state, cirrhosis and hepato-cellular carcinoma.
เลขทะเบียน 1C 7/61 (B)
โปรดอ่านรายละเอียดเพิ่มเติมในเอกสารกำกับยา
ฆศ. 2-6807 / 2568
ความถูกต้องของโฆษณานี้เป็นความรับผิดชอบของผู้โฆษณา มิได้ดำเนินการโดยสำนักงานคณะกรรมการอาหารและยา
เลขทะเบียน 1C 7/61 (B)
โปรดอ่านรายละเอียดเพิ่มเติมในเอกสารกำกับยา
ฆศ. 2-6807 / 2568
ความถูกต้องของโฆษณานี้เป็นความรับผิดชอบของผู้โฆษณา มิได้ดำเนินการโดยสำนักงานคณะกรรมการอาหารและยา
อ่านเพิ่มเติม
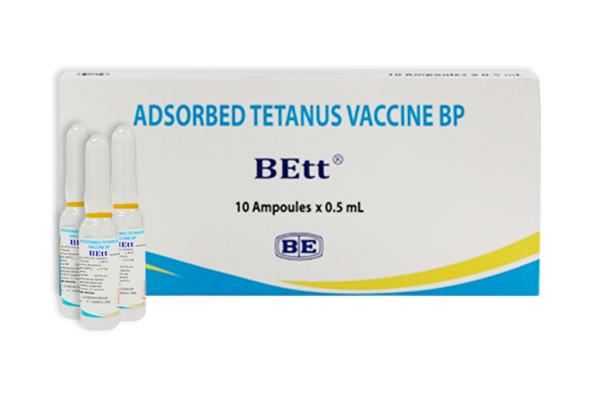
(Adsorbed Tetanus Vaccine)
Indication : Tetanus prophylaxis: Adsorbed Tetanus vaccine is indicated for active immunization against tetanus in adults, children and in infants 6 weeks of age or older. Monovalent Adsorbed Tetanus vaccine should not be used in children less than 7 years of age.
Post exposure prophylaxis of tetanus: Tetanus infection may not confer immunity; therefore initiation or completion of active immunization is indicated at the time of recovery from this infection.
Neonatal tetanus prevention: If vaccination is required, tetanus toxoid can be used during pregnancy. Waiting until the second trimester to administer tetanus vaccine is a reasonable precaution for minimizing any concern regarding the theoretical possibility of adverse reactions.
Tetanus Prophylaxis in Wound Management: Tetanus toxoid can also be used prophylactically for wound management in persons 7 years of age and older.
เลขทะเบียน 1C 15008/62 (NB)
โปรดอ่านรายละเอียดเพิ่มเติมในเอกสารกำกับยา
ฆศ.2-6864/2568
ความถูกต้องของโฆษณานี้เป็นความรับผิดชอบของผู้โฆษณา มิได้ดำเนินการโดยสำนักงานคณะกรรมการอาหารและยา
Post exposure prophylaxis of tetanus: Tetanus infection may not confer immunity; therefore initiation or completion of active immunization is indicated at the time of recovery from this infection.
Neonatal tetanus prevention: If vaccination is required, tetanus toxoid can be used during pregnancy. Waiting until the second trimester to administer tetanus vaccine is a reasonable precaution for minimizing any concern regarding the theoretical possibility of adverse reactions.
Tetanus Prophylaxis in Wound Management: Tetanus toxoid can also be used prophylactically for wound management in persons 7 years of age and older.
เลขทะเบียน 1C 15008/62 (NB)
โปรดอ่านรายละเอียดเพิ่มเติมในเอกสารกำกับยา
ฆศ.2-6864/2568
ความถูกต้องของโฆษณานี้เป็นความรับผิดชอบของผู้โฆษณา มิได้ดำเนินการโดยสำนักงานคณะกรรมการอาหารและยา
อ่านเพิ่มเติม
Tetanus and Diphtheria vaccine
(adsorbed, reduced antigen content)
Indication : The vaccine is indicated for active immunization of children over 7 years of age, adolescents and adults
against tetanus and diphtheria.
เลขทะเบียน 2C 3/57 (B)
โปรดอ่านรายละเอียดเพิ่มเติมในเอกสารกำกับยา
ฆศ. 2-6808 / 2568
ความถูกต้องของโฆษณานี้เป็นความรับผิดชอบของผู้โฆษณา มิได้ดำเนินการโดยสำนักงานคณะกรรมการอาหารและยา
เลขทะเบียน 2C 3/57 (B)
โปรดอ่านรายละเอียดเพิ่มเติมในเอกสารกำกับยา
ฆศ. 2-6808 / 2568
ความถูกต้องของโฆษณานี้เป็นความรับผิดชอบของผู้โฆษณา มิได้ดำเนินการโดยสำนักงานคณะกรรมการอาหารและยา
อ่านเพิ่มเติม
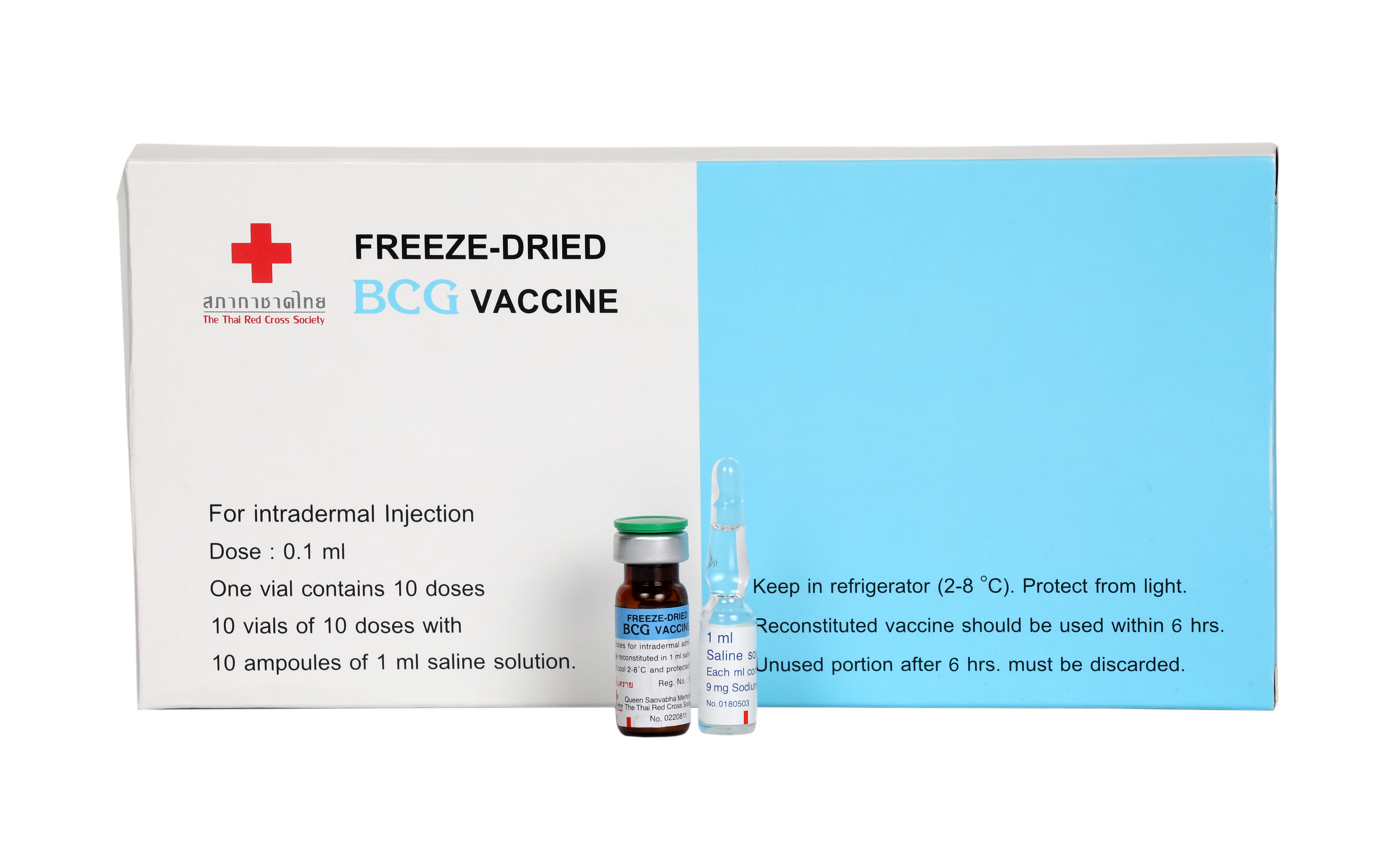
(BCG Vaccine)
Indication : BCG Vaccine is recommended for immunization against tuberculosis
เลขทะเบียน 1A 825/43
โปรดอ่านรายละเอียดเพิ่มเติมในเอกสารกำกับยา
ฆศ. 2-6809 / 2568
ความถูกต้องของโฆษณานี้เป็นความรับผิดชอบของผู้โฆษณา มิได้ดำเนินการโดยสำนักงานคณะกรรมการอาหารและยา
เลขทะเบียน 1A 825/43
โปรดอ่านรายละเอียดเพิ่มเติมในเอกสารกำกับยา
ฆศ. 2-6809 / 2568
ความถูกต้องของโฆษณานี้เป็นความรับผิดชอบของผู้โฆษณา มิได้ดำเนินการโดยสำนักงานคณะกรรมการอาหารและยา
อ่านเพิ่มเติม
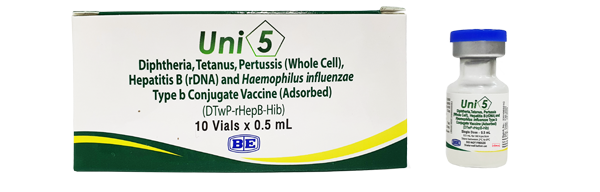
(Diphtheria, Tetanus, Pertussis (Whole cell), Hepatitis B (rDNA) and Haemophilus influenzae Type b Conjugate Vaccine (Adsorbed))
Indication : Uni5 is indicated for active immunization against diphtheria, tetanus, pertussis, hepatitis B (HB) and Haemophilus influenzae type b disease in infants from 6 weeks of age.
เลขทะเบียน 2C 15082/61 (NB)
โปรดอ่านรายละเอียดเพิ่มเติมในเอกสารกำกับยา
ฆศ.2-506/2566
ความถูกต้องของโฆษณานี้เป็นความรับผิดชอบของผู้โฆษณา มิได้ดำเนินการโดยสำนักงานคณะกรรมการอาหารและยา
เลขทะเบียน 2C 15082/61 (NB)
โปรดอ่านรายละเอียดเพิ่มเติมในเอกสารกำกับยา
ฆศ.2-506/2566
ความถูกต้องของโฆษณานี้เป็นความรับผิดชอบของผู้โฆษณา มิได้ดำเนินการโดยสำนักงานคณะกรรมการอาหารและยา
อ่านเพิ่มเติม
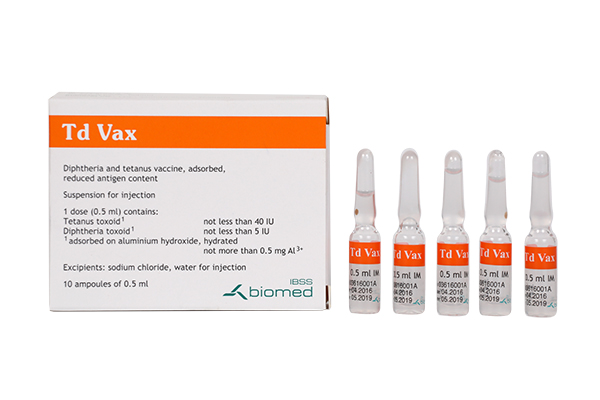
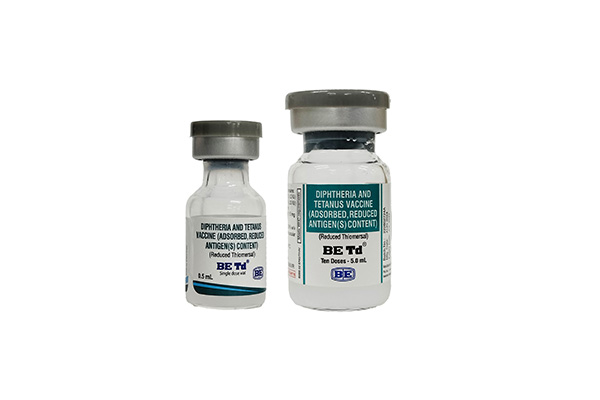
(Diphtheria and Tetanus Vaccine (Adsorbed, Reduced Antigen(s) Content))
Indication : BE Td indicated for active immunization of children 7 years of age or older, and adults, against tetanus and diphtheria.
เลขทะเบียน 2C 3/60 (B)
โปรดอ่านรายละเอียดเพิ่มเติมในเอกสารกำกับยา
ฆศ.2-5499/2568
ความถูกต้องของโฆษณานี้เป็นความรับผิดชอบของผู้โฆษณา มิได้ดำเนินการโดยสำนักงานคณะกรรมการอาหารและยา
เลขทะเบียน 2C 3/60 (B)
โปรดอ่านรายละเอียดเพิ่มเติมในเอกสารกำกับยา
ฆศ.2-5499/2568
ความถูกต้องของโฆษณานี้เป็นความรับผิดชอบของผู้โฆษณา มิได้ดำเนินการโดยสำนักงานคณะกรรมการอาหารและยา
อ่านเพิ่มเติม
(Zoster Virus Vaccine (live) [Oka/SK])
Indication : Prevention of herpes zoster (shingles) in individuals aged 50 years and older.
เลขทะเบียน 1C 19/63 (NBC)
โปรดอ่านรายละเอียดเพิ่มเติมในเอกสารกำกับยา
ฆศ.2-4063/2568
ความถูกต้องของโฆษณานี้เป็นความรับผิดชอบของผู้โฆษณา มิได้ดำเนินการโดยสำนักงานคณะกรรมการอาหารและยา
เลขทะเบียน 1C 19/63 (NBC)
โปรดอ่านรายละเอียดเพิ่มเติมในเอกสารกำกับยา
ฆศ.2-4063/2568
ความถูกต้องของโฆษณานี้เป็นความรับผิดชอบของผู้โฆษณา มิได้ดำเนินการโดยสำนักงานคณะกรรมการอาหารและยา
อ่านเพิ่มเติม
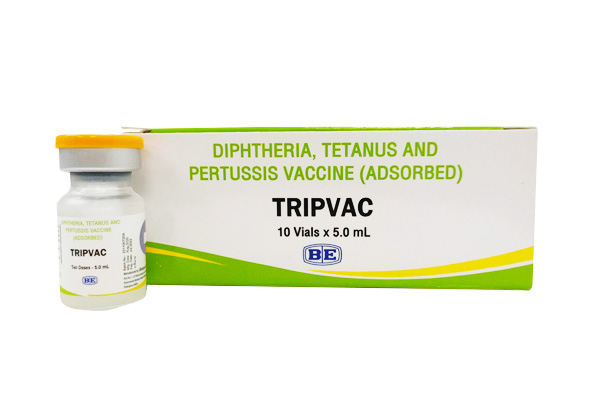
Tripvac
(Diphtheria, Tetanus and Pertussis Vaccine (Adsorbed))
Indication : Indication: Diphtheria, Tetanus and Pertussis Vaccine (Adsorbed) (DTwP) is indicated for primary immunization of infants, above the age of six weeks against diphtheria, tetanus and whooping cough diseases. The vaccine can be safely and effectively given at the same time as BCG, Measles, Polio (OPV and IPV), Hepatitis B, Yellow fever, Haemophilus influenzae type b vaccines and Vitamin A supplementation.
เลขทะเบียน 2C 15018/62 (NB)
โปรดอ่านรายละเอียดเพิ่มเติมในเอกสารกำกับยา
ฆศ.2-269/ 2569
ความถูกต้องของโฆษณานี้ เป็นความรับผิดชอบของผู้โฆษณา มิได้ดำเนินการโดยสำนักงานคณะกรรมการอาหารและยา
เลขทะเบียน 2C 15018/62 (NB)
โปรดอ่านรายละเอียดเพิ่มเติมในเอกสารกำกับยา
ฆศ.2-269/ 2569
ความถูกต้องของโฆษณานี้ เป็นความรับผิดชอบของผู้โฆษณา มิได้ดำเนินการโดยสำนักงานคณะกรรมการอาหารและยา
อ่านเพิ่มเติม